A new route for the synthesis of PalbociclibShu-ting Li, Jun-qing Chen, Cheng-liang Feng, Wan-feng Yang, and Min Ji Southeast University, Nanjing, People’s Republic of China
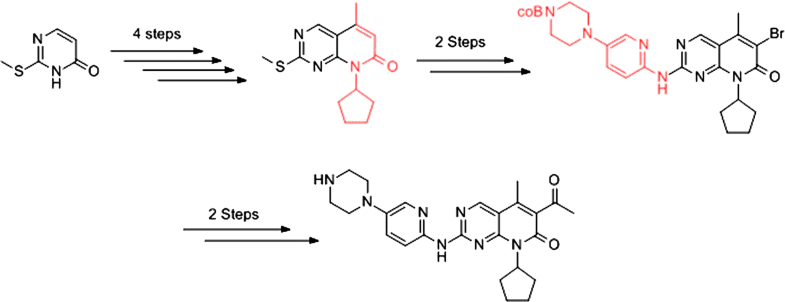
E-mail: jqchen@seu.edu.cn Abstract: In this paper, a novel synthetic method for Palbociclib was reported. It was synthesized in eight steps from 2-(methylthio) pyrimidin-4-(3H)-one with approximately 10% overall yield. This protocol started material 2-(methylthio) pyrimidin-4-(3H)-one, involved nucleophilic substitution by thionyl chloride, bromination, nucleophilic substitution by cyclopentylamine, a one pot-two step method (Heck reaction, ring close sequence), oxidation and bromination, cross-coupling reaction, Heck reaction, aqueous workup to afford Palbociclib. This synthetic route used inexpensive raw material and reagents, involved readily controllable reaction conditions and reduced environmental hazards. Synthesis of Palbociclib, a small molecule CDK inhibitor, starting from 2-(methylthio) pyrimidin-4-(3H)-one by 8 steps reaction. This method afforded the Palbociclib in 10% yield. Keywords: CDK ; CDK inhibitor ; Palbociclib ; Synthesis Full paper is available at www.springerlink.com. DOI: 10.1007/s11696-019-00841-7
Chemical Papers 73 (12) 3043–3051 (2019) |
Friday, May 01, 2026 |
|||
© 2026 Chemical Papers |
||||