Study of ciprofloxacin degradation by zero-valent copper nanoparticlesPaloma Viana Ferreira de Sousa, André Fernando de Oliveira, Antônio Alberto da Silva, Boniek Gontijo Vaz, and Renata Pereira Lopes Federal University of Viçosa, Viçosa, Brazil
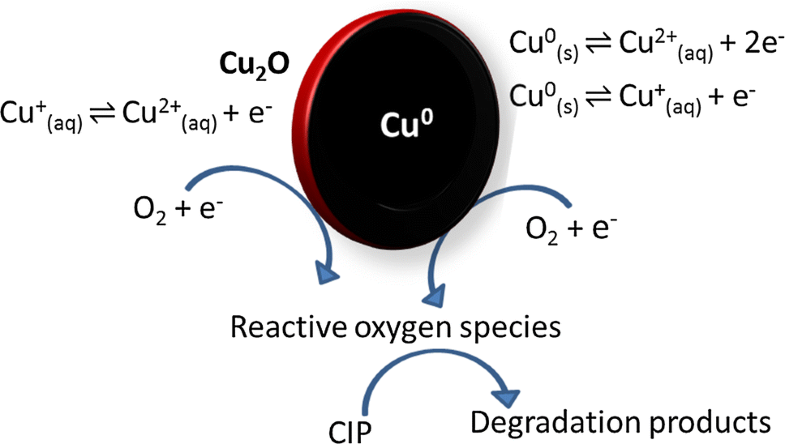
E-mail: renata.plopes@ufv.br Abstract: In this study, ciprofloxacin (CIP) degradation by zero-valent copper nanoparticles (nZVC) was investigated. The nZVC were characterized by transmission electron microscopy (TEM), X-ray spectroscopy by dispersion in energy (EDS) and X-ray diffraction (XRD). Approximately 100% of degradation was obtained at CIP concentration of 20 mg L−1, pH 3.5 and dose of nZVC of 0.5 g L−1. The reaction mechanism was investigated and showed that the CIP degradation occurs in acidic conditions via active radical-forming species of oxygen, formed from Cu(I). In basic conditions the removal mechanism proved to be different from the one observed for the acidic conditions. The desorption studies confirmed that the CIP is adsorbed in the nZVC in this condition. The degradation kinetics is favored by the increased dosage and temperature and the addition of chloride anion. The nanoparticles reuse assays (a cycle) were performed and showed an efficiency of about 70%. The residual copper (50 mg L−1) in the system was minimized by precipitation assays at pH 8.8, showing a concentration of only 1.05 mg L−1. Finally, proposals were made of the degradation by-products, which indicated the molecule oxidation, proving the degradation hypothesis via active radical-forming oxygen species. Keywords: Zero-valent copper ; Nanoparticles ; Ciprofloxacin ; Degradation Full paper is available at www.springerlink.com. DOI: 10.1007/s11696-018-0575-7
Chemical Papers 73 (1) 249–260 (2019) |
Friday, April 19, 2024 |
|||
© 2024 Chemical Papers |
||||