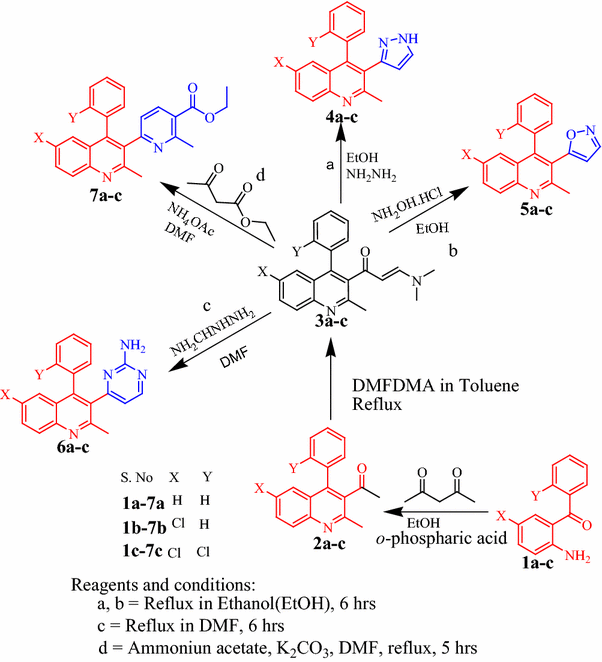
An efficient solvent-free synthesis of 3-acetyl-4-arylquinoline-based enaminones and its derivatives using DMFDMA reagentL. Jyothish Kumar and V. Vijayakumar Centre for Organic and Medicinal Chemistry, School of Advanced Sciences, VIT University, Vellore, India
E-mail: kvpsvijayakumar@gmail.com Abstract: A series of 3-substituted-4-arylquinoline derivatives were synthesized using 3-acetyl-4-arylquinoline. The acetyl function of 3-acetyl-4-arylquinoline was successfully converted into its corresponding enaminone using DMFDMA as a reagent which in turn successfully converted into pyrazole, isoxazole, pyrimidine, phenyl aminoprop-2-en-1-one, pyridin-2-yl-amino-prop-2-en-1-one, 2-methylpyridine-3-carboxylate by treating with reagents such as hydrazine, hydroxylamine, guanidine hydrochloride, aniline, 2-amino pyridine, ethyl acetoacetate, respectively, under solvent-free microwave irradiation as well as under the conventional thermal heating processes. All the synthesized compounds were found to be obtained in better yields under the microwave irradiation over the conventional process. Keywords: DMFDMA ; Enaminones ; Microwave irradiation ; Solvent-free conditions Full paper is available at www.springerlink.com. DOI: 10.1007/s11696-017-0375-5
Chemical Papers 72 (8) 2001–2012 (2018) |
Friday, May 01, 2026 |
|||
© 2026 Chemical Papers |
||||